Molecular biologists and biochemists are always talking about their sample’s 260/280 ratio. It is so commonly used that entire instruments are dedicated to displaying this value when analyzing a sample. The A260/A280 ratio is a procedure that tells scientists the extinct of contamination of their nucleic acid solution by proteins, carbohydrates, and other organic molecules.
The basis of this test relies on the Beer-Lambert law: A = εbc; where A is absorbance, e is the molar extinction coefficient, b is the cell path length, and c is the sample concentration. The commonly accepted average extinction coefficients for a 1 mg/mL nucleic acid solution at 260 nm and 280 nm are 20 and 10, respectively. In proteins, the extinction coefficient values at 260 nm and 280 nm at a concentration of 1 mg/mL are 0.57 and 1.00, respectively. Therefore, nucleic acid samples would be expected to have a higher absorbance at 260 nm than at 280 nm; in a protein sample, the opposite is true. Using these extinction coefficients, pure nucleic acid samples would have an A260/A280 ratio of 2.0, while protein would be 0.57.
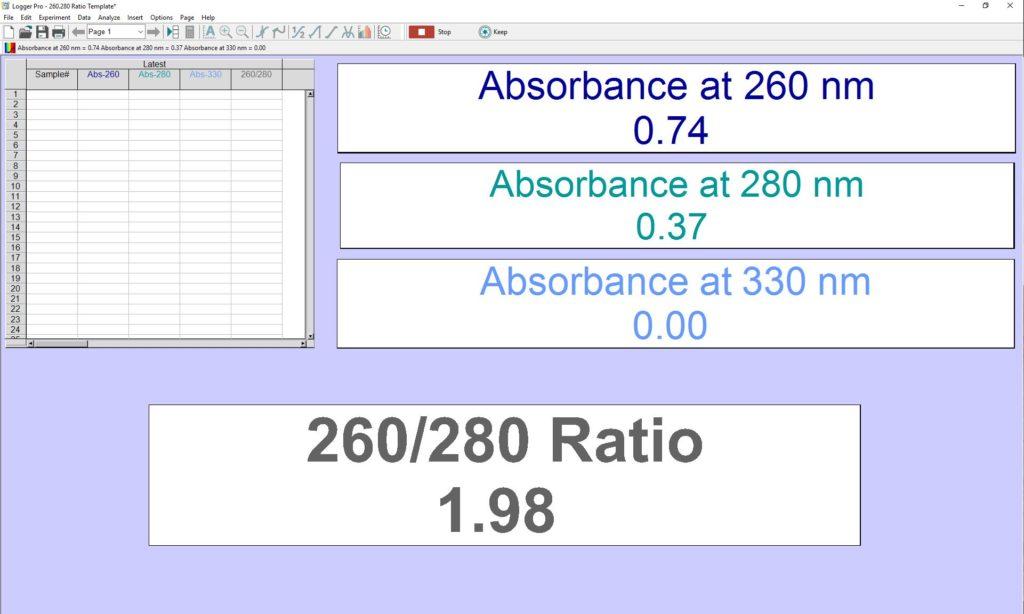
In order to make the determination of a 260/280 ratio easier with a Vernier UV-VIS Spectrophotometer or a Vernier Fluorescence/UV-VIS Spectrophotometer, we have developed a Logger Pro™ 3 template that you can use to easily see the ratio without having to go through the entire process every time. A screenshot of the Logger Pro template is shown below.
To use this template, first download the template file. Connect the spectrometer to your computer and open the file in Logger Pro. After you have properly calibrated your spectrometer and prepped your sample, insert your sample into the spectrometer. Press the Collect button and the A260/A280 ratio value will be displayed live in the meter. The table is also displayed in the Logger Pro template. If you double click on the 260/280 header in the table, you can see the calculations used to generate the value and make modifications, if desired.
Vernier has great options for biochemistry, be sure to visit our biochemistry solution page for more free resources and great tips.
Download our Logger Pro template for completing a 260/280 ratio »