Evaporation and Intermolecular Attractions
Experiment #8 from Investigating Chemistry through Inquiry
- Subject
- Chemistry
Introduction
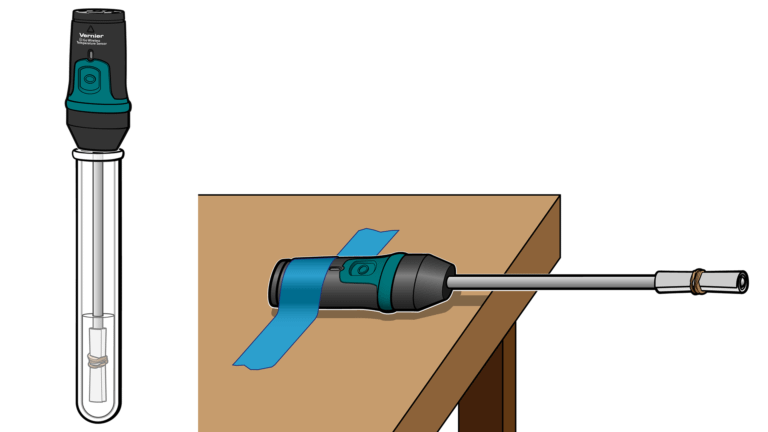
The evaporation of a volatile liquid from the surface of a Temperature Probe is an endothermic process that results in a temperature decrease. The magnitude of a temperature decrease is, like viscosity and boiling temperature, related to the strength of intermolecular forces of attraction. In this experiment, you will investigate factors that determine strengths of intermolecular forces of attraction in alkanes and alcohols and the magnitudes of the temperature changes as these substances evaporate from a Temperature Probe.
Objectives
In the Preliminary Activity, you will gain experience using a Temperature Probe while you determine the temperature change as ethanol, C2H5OH, evaporates.
After completing the Preliminary Activity, you will first use reference sources to find out more about alkanes and alcohols before you choose and investigate a researchable question dealing with temperature changes as alkanes and alcohols evaporate.
Sensors and Equipment
This experiment features the following sensors and equipment. Additional equipment may be required.
Option 1
Ready to Experiment?
Ask an Expert
Get answers to your questions about how to teach this experiment with our support team.
- Call toll-free: 888-837-6437
- Chat with Us
- Email support@vernier.com
Purchase the Lab Book
This experiment is #8 of Investigating Chemistry through Inquiry. The experiment in the book includes student instructions as well as instructor information for set up, helpful hints, and sample graphs and data.


