For so many regions, autumn brings dramatic color changes to deciduous forests. The reds, oranges, and yellows of autumn provide inspiration for paintings and calendars, but why do these dramatic color changes occur? What does science have to say about this phenomenon?
We have a free experiment that challenges biology students to use data-collection technology to better understand why a tree’s leaves change colors. Since many campuses remain committed to remote learning through the first term, we’ve included free sample data for students to use as they study from home.
The Science Behind Autumn Colors
As day lengths grow shorter as summer turns to autumn, many trees slow their production of chlorophyll, while other pigments such as anthocyanin may be produced in higher quantities. Leaves shift from green to red, orange, or yellow before finally dropping from the tree.
To better understand this change in leaves, students can use spectroscopy to study pigments from tree leaves of different colors and compare absorption spectra.
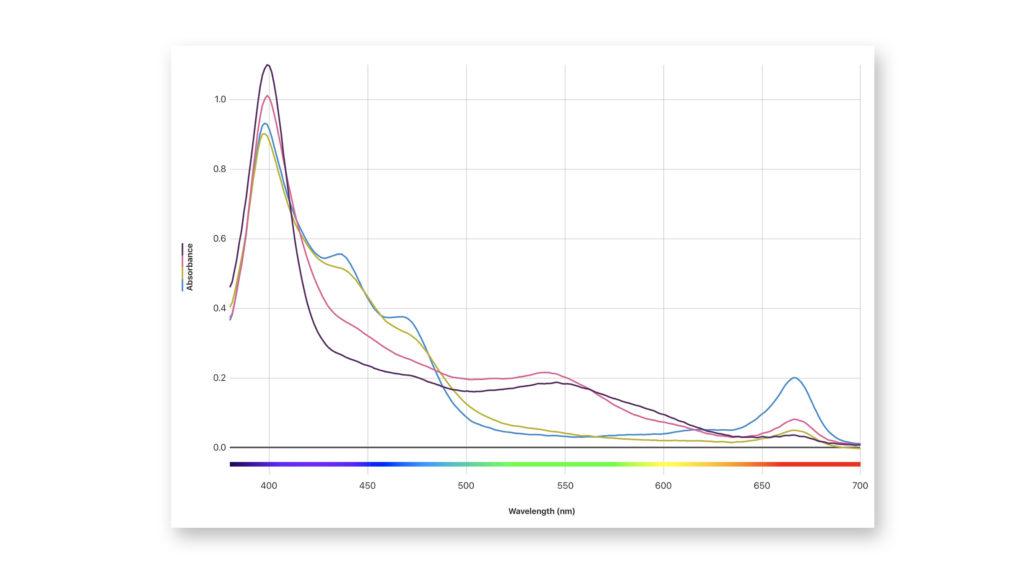
Using the Vernier Spectral Analysis® app, students can analyze the sample data provided as a download at the bottom of this post. Spectral Analysis is a free app designed to help students collect and analyze spectrometer data. This app is compatible with Windows®, macOS®, Chromebook™, iOS, iPadOS®, and Android™ systems, which means students can use the app to analyze on virtually any device, no matter where they are.
In our “Visible Spectra of Plant Pigments” experiment from Advanced Biology with Vernier, students soak plant material in 70% isopropyl alcohol to create pigment extracts and then measure their absorbance spectra using a spectrometer. While the experiment calls for spinach leaves, it is easy to substitute colored fall leaves if available in your area (collect them fresh from the tree for best results). Shredding and grinding the leaves helps release more pigments for the extract, and soaking time can be extended, if necessary.
Important note: The experiment, as written, includes instructions involving acetone and petroleum. To make this experiment safe for students studying from home, omit the steps involving acetone and petroleum ether, since these substances should only be used in a fume hood.
Download “Visible Spectra of Plant Pigments”
Students will finish this experiment with valuable data-collection experience and a deeper understanding of how spectroscopy works. If the leaves have already turned in your area, don’t worry—you can substitute autumn leaves with cranberries, spinach, or pumpkin.