Properties of Solutions: Electrolytes and Non-Electrolytes
Experiment #13 from Chemistry with Vernier
- Education Level
- High School
- Subject
- Chemistry
Introduction
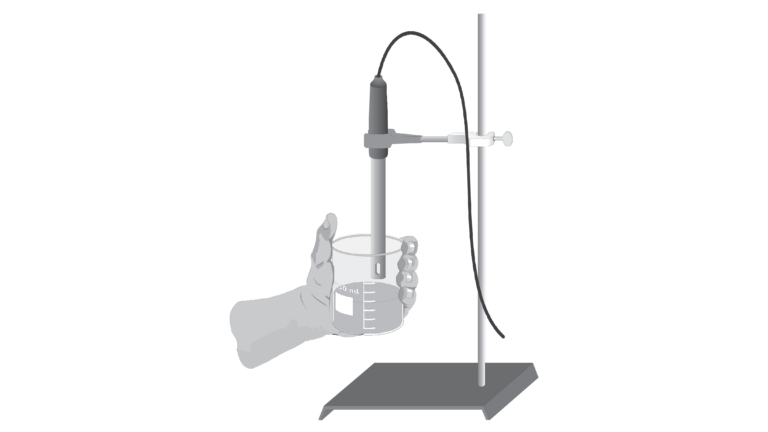
In this experiment, you will discover some properties of strong electrolytes, weak electrolytes, and non-electrolytes by observing the behavior of these substances in aqueous solutions. You will determine these properties using a Conductivity Probe. When the probe is placed in a solution that contains ions, and thus has the ability to conduct electricity, an electrical circuit is completed across the electrodes that are located on either side of the hole near the bottom of the probe body. This results in a conductivity value that can be read by the computer. The unit of conductivity used in this experiment is the microsiemens per centimeter, or μS/cm.
The size of the conductivity value depends on the ability of the aqueous solution to conduct electricity. Strong electrolytes produce large numbers of ions, which results in high conductivity values. Weak electrolytes result in low conductivity, and non-electrolytes should result in no conductivity. In this experiment, you will observe several factors that determine whether or not a solution conducts, and if so, the relative magnitude of the conductivity. Thus, this simple experiment allows you to learn a great deal about different compounds and their resulting solutions.
Objectives
In this experiment, you will
- Write equations for the dissociation of compounds in water.
- Use a Conductivity Probe to measure the conductivity of solutions.
- Determine which molecules or ions are responsible for conductivity of solutions.
- Investigate the conductivity of solutions resulting from compounds that dissociate to produce different numbers of ions.
Sensors and Equipment
This experiment features the following sensors and equipment. Additional equipment may be required.
Option 1

Option 2

Correlations
Teaching to an educational standard? This experiment supports the standards below.
- International Baccalaureate (IB) 2025/Chemistry
- Structure 2.1.2—The ionic bond is formed by electrostatic attractions between oppositely charged ions.
Ready to Experiment?
Ask an Expert
Get answers to your questions about how to teach this experiment with our support team.
- Call toll-free: 888-837-6437
- Chat with Us
- Email support@vernier.com
Purchase the Lab Book
This experiment is #13 of Chemistry with Vernier. The experiment in the book includes student instructions as well as instructor information for set up, helpful hints, and sample graphs and data.

