Introduction
Food supplies energy for all animals—without it we could not live. The quantity of energy stored in food is of great interest to humans. The energy your body needs for running, talking, and thinking comes from the foods you eat. Not all foods contain the same amount of energy, nor are all foods equally nutritious for you. An average person should consume a minimum of 2,000 kilocalories per day. That is equivalent to 8,360 kilojoules. Calories and joules are both units of energy. We will use joules in this lab since it is the accepted SI metric standard.
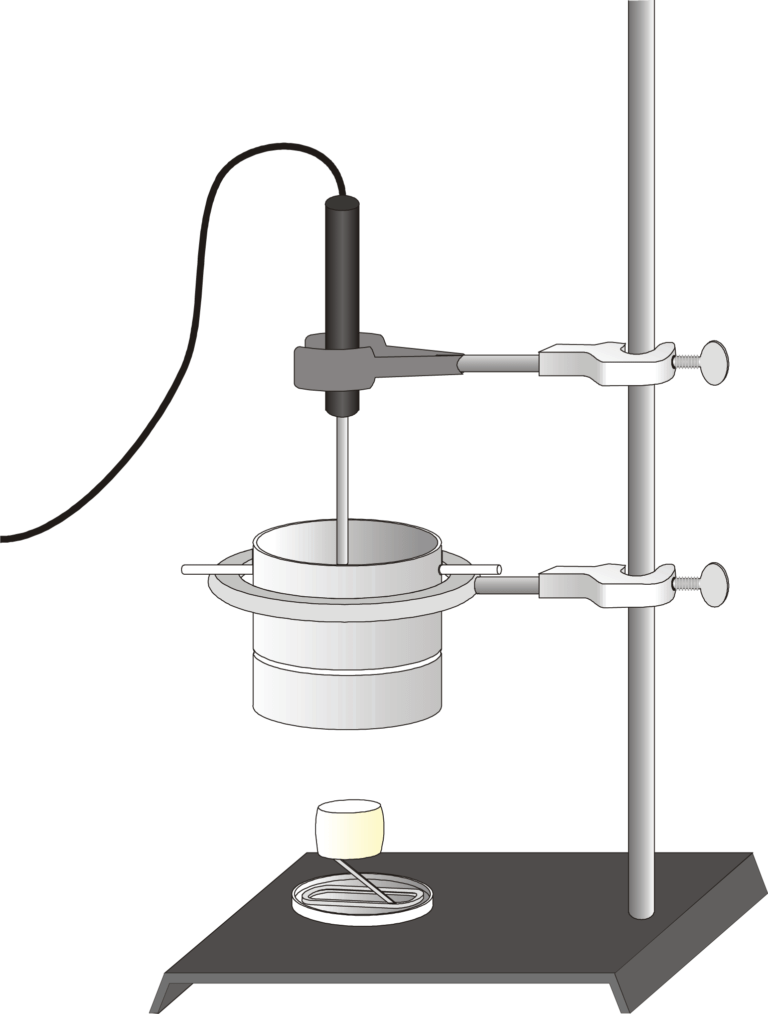
You can determine energy content of food by burning a portion of it and capturing the heat released to a known amount of water. This technique is called calorimetry. The energy content of the food is the amount of heat produced by the combustion of 1 gram of a substance. It is measured in kilojoules per gram (kJ/g).
Objectives
In this experiment, you will
- Measure temperature changes.
- Monitor the energy given off by food as it burns.
- Determine and compare the energy content of different foods.
Sensors and Equipment
This experiment features the following sensors and equipment. Additional equipment may be required.
Option 1

Correlations
Teaching to an educational standard? This experiment supports the standards below.
- International Baccalaureate (IB)/Sports, Exercise, and Health Science
- 3.1 Nutrition
Ready to Experiment?
Ask an Expert
Get answers to your questions about how to teach this experiment with our support team.
- Call toll-free: 888-837-6437
- Chat with Us
- Email support@vernier.com
Purchase the Lab Book
This experiment is #1 of Biology with Vernier. The experiment in the book includes student instructions as well as instructor information for set up, helpful hints, and sample graphs and data.


