Fractional Distillation of Esters
Experiment #9 from Organic Chemistry with Vernier
- Education Level
- College
Introduction
Fractional distillation is a technique used to separate mixtures of compounds into pure components. At a given temperature, a pure liquid has a specific vapor pressure. Heating the liquid provides a greater mole fraction of molecules with the kinetic energy needed to escape into the gas phase. This temperature, when the vapor pressure of a liquid equals the atmospheric pressure, is defined as the boiling temperature of a substance.
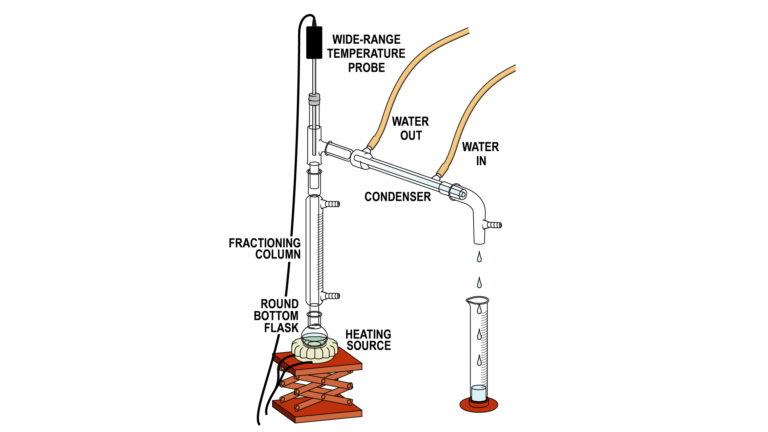
If substances in a mixture have significantly different vapor pressures, they can be separated by a technique known as simple distillation. In simple distillation, a mixture is warmed slowly and the most volatile liquid vaporizes first. The vapor is then condensed back to liquid and collected in a separate flask. For mixtures of compounds with similar boiling points, you could perform a simple distillation multiple times, or employ fractional distillation.
A fractionating column is one designed to allow multiple simple distillations to occur over its length. One type of fractionating column is a long glass tube packed with glass or ceramic beads that allows the vapor to condense and vaporize multiple times as it travels up the column. Each cycle of condensation and evaporation is called a theoretical plate. The more theoretical plates in the column, the better the separation between the compounds in a mixture will be.
Objectives
In this experiment, you will
- Measure and analyze the retention times and peak areas of ethyl acetate and butyl acetate.
- Conduct the fractional distillation of a mixture of ethyl acetate and butyl acetate.
- Measure and analyze the retention times and peak areas of the fractions.
- Estimate the mole fraction of the components of the mixture.
Sensors and Equipment
This experiment features the following sensors and equipment. Additional equipment may be required.
Ready to Experiment?
Ask an Expert
Get answers to your questions about how to teach this experiment with our support team.
- Call toll-free: 888-837-6437
- Chat with Us
- Email support@vernier.com
Purchase the Lab Book
This experiment is #9 of Organic Chemistry with Vernier. The experiment in the book includes student instructions as well as instructor information for set up, helpful hints, and sample graphs and data.




