Soil and Acid Rain
Experiment #12 from Investigating Environmental Science through Inquiry
- Subject
- Environmental Science
Introduction
Acid precipitation can be very harmful to the environment. It can kill fish by lowering the pH of lakes and rivers. It can harm trees and plants by burning their leaves and depriving them of nutrients. In addition, it can weather away stone buildings and monuments.
Carbon dioxide, CO2, is a gas found naturally in the air. When CO2 dissolves into water, it produces a weak acid called carbonic acid, H2CO3. This makes precipitation slightly acidic naturally. Precipitation of pH 5 to 6 is common and does not generally cause any problems. Oxides of sulfur and nitrogen released into the air by fossil fuel burning power plants, various industries, and automobiles dissolve into atmospheric water to form acids such as H2SO3 (sulfurous acid), H2SO4 (sulfuric acid), HNO2 (nitrous acid), and HNO3 (nitric acid). The resulting precipitation can be as acidic as pH 4, precipitation with a pH below 5.6 is generally considered to be “acid precipitation.”
Objectives
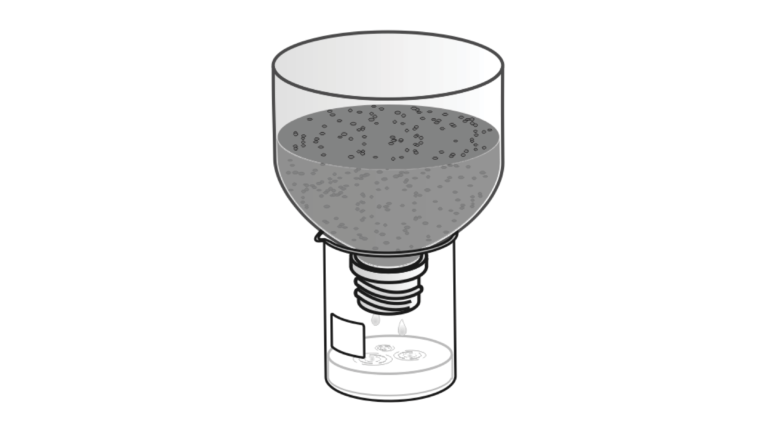
In the Preliminary Activity, you will use a pH Sensor to measure the pH of acid rainwater. You will then filter the acid rainwater through soil, collect the resulting filtrate, and measure the filtrate pH as you determine the soil’s influence on acid rainwater pH.
After completing the Preliminary Activity, you will first use reference sources to find out more about acid precipitation, soil pH, and soil buffering capacity before you choose and investigate a researchable question.
Sensors and Equipment
This experiment features the following sensors and equipment. Additional equipment may be required.
Correlations
Teaching to an educational standard? This experiment supports the standards below.
- International Baccalaureate (IB)/Environmental Systems and Societies
- 6.4 Acid deposition
Ready to Experiment?
Ask an Expert
Get answers to your questions about how to teach this experiment with our support team.
- Call toll-free: 888-837-6437
- Chat with Us
- Email support@vernier.com
Purchase the Lab Book
This experiment is #12 of Investigating Environmental Science through Inquiry. The experiment in the book includes student instructions as well as instructor information for set up, helpful hints, and sample graphs and data.


