Experiments
Here are experiments our science specialists have selected to support this IB* topic.
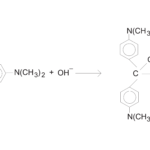
Rate Law Determination of the Crystal Violet Reaction
Experiment #30 from Chemistry with Vernier
In this experiment, you will
- Observe the reaction between crystal violet and sodium hydroxide.
- Monitor the absorbance of the crystal violet solution with time.
- Graph Absorbance vs. time, ln Absorbance vs. time, and 1/Absorbance vs. time.
- Determine the order of the reaction.
- Determine the rate constant, k, and the half-life for this reaction.
* The IB Diploma Program is an official program of the International Baccalaureate Organization (IBO) which authorizes schools to offer it. The material available here has been developed independently of the IBO and is not endorsed by it.
