Introduction
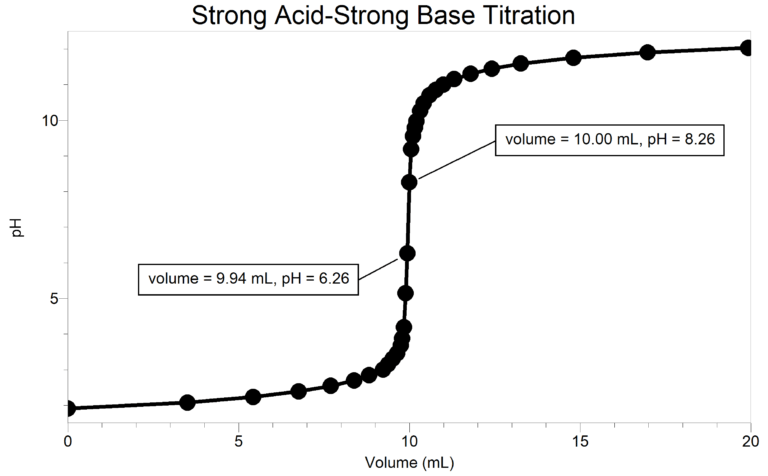
A titration is a process used to determine the volume of a solution needed to react with a given amount of another substance. When titrating a solution of the strong acid hydrochloric acid, HCl, with a solution of the strong base sodium hydroxide, NaOH, the hydrogen ions from the HCl react with hydroxide ions from the NaOH in a one-to-one ratio to produce water in the overall reaction:
When an HCl solution is titrated with an NaOH solution, the pH of the acidic solution is initially low. As base is added, the change in pH is quite gradual until close to the equivalence point, when equimolar amounts of acid and base have been mixed. Near the equivalence point, the pH increases very rapidly. The change in pH then becomes more gradual again, before leveling off with the addition of excess base.
Objectives
In the Preliminary Activity, you will titrate a solution of the strong acid hydrochloric acid, HCl, with a solution of the strong base sodium hydroxide, NaOH. The concentration of the NaOH solution is given and you will determine the unknown concentration of the HCl.
After completing the Preliminary Activity, you will first use reference sources to find out more about acids, bases, and acid-base titrations before you choose and investigate a researchable question utilizing acid-base titrations.
Sensors and Equipment
This experiment features the following sensors and equipment. Additional equipment may be required.
Option 1

Option 2

Ready to Experiment?
Ask an Expert
Get answers to your questions about how to teach this experiment with our support team.
- Call toll-free: 888-837-6437
- Chat with Us
- Email support@vernier.com
Purchase the Lab Book
This experiment is #17 of Investigating Chemistry through Inquiry. The experiment in the book includes student instructions as well as instructor information for set up, helpful hints, and sample graphs and data.

