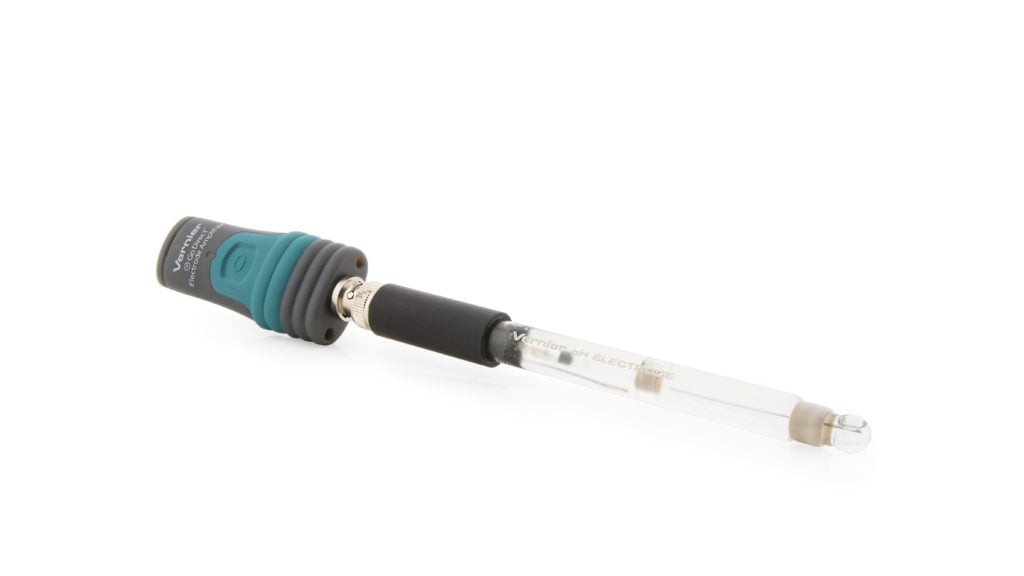
Go Direct® Glass-Body pH Sensor User Manual
Order Code: GDX-GPH
The Go Direct Glass-Body pH is a high-quality glass-body pH sensor that can be used in non-aqueous solutions that contain organic solvents and solutions that contain strong acids or strong bases.
Note: Vernier products are designed for educational use. Our products are not designed nor are they recommended for any industrial, medical, or commercial process such as life support, patient diagnosis, control of a manufacturing process, or industrial testing of any kind.
What's Included
- Go Direct Glass-Body pH (Go Direct Electrode Amplifier connected to a Go Direct Glass-Body pH BNC Electrode)
- Electrode storage bottle, containing pH 4/KCl solution
- Micro USB Cable
- Electrode tip guard
Compatible Software
Choose a platform below to see its compatibility requirements.LabQuest
Interface LabQuest App LabQuest 3 Full support LabQuest 2 (discontinued) Full support 1 LabQuest (discontinued) Incompatible Compatibility Notes
Computers
Software Interface Graphical Analysis No interface required Full support 1 LabQuest 3 Full support 2 LabQuest 2 (discontinued) Full support 2 3 Compatibility Notes
Chromebook
Software Interface Graphical Analysis No interface required Full support LabQuest 3 Full support 1 LabQuest 2 (discontinued) Full support 1 2 Compatibility Notes
iOS
Software Interface Graphical Analysis Graphical Analysis GW No interface required Full support Full support LabQuest 3 Full support 1 2 Full support 1 2 LabQuest 2 (discontinued) Full support 1 2 3 Full support 1 2 3 Compatibility Notes
Android
Software Interface Graphical Analysis Graphical Analysis GW No interface required Full support Incompatible LabQuest 3 Full support 1 2 Full support 1 LabQuest 2 (discontinued) Full support 1 2 3 Full support 1 3 Compatibility Notes
Python
Software Interface Python No interface required Full support Javascript
Software Interface Javascript No interface required Full support 1 Compatibility Notes
LabVIEW
Software Interface NI LabVIEW No interface required Full support 1 Compatibility Notes
Getting Started
Please see the following link for platform-specific connection information:
|
Bluetooth Connection |
USB Connection |
|
|
Charging the Sensor
Connect the Go Direct Glass-Body pH to the included Micro USB Cable and any USB device for two hours. Connecting the Go Direct pH BNC Electrode to the electrode amplifier during charging is optional.
You can also charge up to eight Go Direct Glass-Body pHs using our Go Direct Charge Station, sold separately (order code: GDX-CRG). An LED on each Go Direct Glass-Body pH indicates charging status.
| Charging |
Blue LED on steady while sensor is connected to the Micro USB Cable or Charge Station. |
| Fully charged |
Blue LED is off when charging is complete. |
Powering the Sensor
| Turning on the sensor |
Press button once. Red LED indicator flashes when unit is on. |
| Putting the sensor in sleep mode |
Press and hold button for more than three seconds to put into sleep mode. Red LED indicator stops flashing when sleeping. |
Connecting the Sensor
See the following link for up-to-date connection information:
| Connected and charging | Blue and Green LED solid when sensor is connected to Graphical Analysis via USB and unit is charging. (Green LED is obscured by the blue one.) |
| Connected, fully charged | Green LED solid when sensor is connected to Graphical Analysis via USB and the unit is fully charged. |
| Charging via USB, connected via Bluetooth |
Blue LED is solid and green LED is flashing, but the green flashing LED looks white because it is overwhelmed by the blue. |
Identifying the Sensor
When two or more sensors are connected, the sensors can be identified by tapping or clicking Identify in Sensor Information.
Using the Product
- Remove the storage bottle from the electrode by unscrewing the lid and removing the bottle and lid.
- Thoroughly rinse the lower section of the probe, especially around the bulb shaped tip, using distilled or deionized water.
- Connect the sensor following the steps in the Getting Started section of this user manual.
- When you are finished making measurements, rinse the electrode with distilled water.
- Slide the cap onto the electrode body, and then screw the cap onto the storage bottle so the tip of the electrode is immersed in the storage solution.
Important: Do not fully submerge the sensor. The BNC connection is not waterproof.
By default, the units displayed will be in pH. See www.vernier.com/til/3984 to change the displayed units.
Calibrating the Sensor
For many experiments, calibrating the Go Direct Glass-Body pH is not required. A calibration equation is stored on each pH Sensor before it is shipped, which is used as a default by Vernier software.
For the most accurate measurements with this sensor, we recommend calibration. It is a simple process that takes only a few minutes. For additional calibration information, see www.vernier.com/til/4011
To calibrate the sensor in Graphical Analysis, complete the following steps.
- Click or tap the sensor meter to view sensor options.
- Select Calibrate and follow the applicable prompts on the Calibrate Sensor screen.
In order to calibrate a pH Sensor, or to confirm that a saved pH calibration is accurate, you should have a supply of pH buffer solutions that cover the range of the pH values you will be measuring. For more information about pH buffer solutions, including recipes for preparation, see www.vernier.com/til/3625
Once you have calibrated your Go Direct Glass-Body pH, the calibration is automatically stored to the sensor and will be used each time you connect to your device. You can always choose to restore factory defaults if you feel the custom calibration is invalid.
To restore factory defaults in Graphical Analysis, complete the following steps.
- Click or tap the live readouts meter and choose Calibrate.
- Click or tap Reset Calibration.
- A window will appear warning you that you are about to reset the calibration. Choose Reset Calibration.
Specifications
|
Type |
Sealed, gel-filled, glass body, Ag/AgCl reference |
|
Temperature range |
5 to 80°C (readings not compensated) |
|
Range |
pH 0–14 |
|
Typical accuracy (factory calibration) |
± 0.2 pH units |
|
Isopotential pH |
pH 7 (point at which temperature has no effect) |
|
Shaft diameter |
12 mm OD |
|
Resolution |
0.01 pH |
|
USB specification |
2.0 |
|
Wireless specification |
Bluetooth 4.2 |
|
Maximum wireless range |
30 m |
|
Dimensions |
|
|
Battery |
300 mA Li-Poly |
|
Battery life (single full charge) |
~24 hours |
|
Battery life (long term) |
~500 full charge cycles (several years depending on usage) |
Care and Maintenance
Short-term storage (up to 24 hours): Place the electrode in pH 4 or pH 7 buffer solution. It should never be stored in distilled water.
Long-term storage (more than 24 hours): Store the electrode in a pH 4 buffer/KCl storage solution in the storage bottle. The pH electrode is shipped in this solution. Vernier sells 500 mL bottles of pH Storage Solution (order code PH‑SS), or you can prepare additional storage solution by adding 10 g of solid potassium chloride (KCl) to 100 mL of pH 4 buffer solution. Vernier sells a pH Buffer Capsule kit (PH‑BUFCAP) that includes a buffer solution preservative. Storing the electrode in this solution contributes to electrode longevity and retains electrode response time when the unit is placed back into service.
If the electrode is inadvertently stored dry for a short period of time, immerse the tip in the pH 4 buffer/KCl storage solution for a minimum of 8 hours prior to use. If the readings are still not accurate after calibration or if the response is slow, try shocking the sensor as described in the Troubleshooting section.
Battery Information
The Go Direct Glass-Body pH contains a small lithium-ion battery in the handle. The system is designed to consume very little power and not put heavy demands on the battery. Although the battery is warranted for one year, the expected battery life should be several years. Replacement batteries are available from Vernier (order code: GDX-BAT-300).
Storage and Maintenance
To store the Go Direct Glass-Body pH for extended periods of time, put the device in sleep mode by holding the button down for at least three seconds. The red LED will stop flashing to show that the unit is in sleep mode. Over several months, the battery will discharge but will not be damaged. After such storage, charge the device for a few hours, and the unit will be ready to go.
Exposing the battery to temperatures over 35°C (95°F) will reduce its lifespan. If possible, store the device in an area that is not exposed to temperature extremes.
Water Resistance
The Go Direct Glass-Body pH is not water resistant and should never be immersed in water.
If water gets into the device, immediately power the unit down (press and hold the power button for more than three seconds). Disconnect the sensor and charging cable, and remove the battery. Allow the device to dry thoroughly before attempting to use the device again. Do not attempt to dry using an external heat source.
How the Sensor Works
pH is measured potentiometrically when using an electrode. In other words, your measurement is based on an electrical signal. A potential develops across the glass membrane of a pH electrode when it comes in contact with a solution. This potential varies as the pH varies, but requires a constant second potential to compare the changes against. This is the function of the reference electrode, to provide a constant potential, regardless of pH.
In acidic or alkaline solutions, the voltage on the outer membrane surface changes proportionally to changes in hydrogen ion activity by the Nernst equation:
E = Eo + (2.3RT/nF) log [H+]
Where:
E = total potential difference (measured in mV)
Eo = standard potential
R = gas constant
T = temperature in Kelvin
N = number of electrons
F = Faraday's constant
[H+] = hydrogen ion activity
Troubleshooting
When testing a Go Direct Glass-Body pH, it is best to measure a buffer solution because it is easier to determine if the sensor is reading correctly. Do not test your sensor by measuring distilled water. Distilled water can have a pH reading in the range of 5.5–7.0, due to varying amounts of dissolved carbon dioxide. Furthermore, due to a lack of ions, the pH values reported with the sensor in distilled water will be erratic.
If your pH Sensor is reading differently from the pH of a buffer solution (e.g., reads 6.7 in a buffer 7), you may simply need to calibrate the sensor. See the Calibrating the Sensor section for more information.
Examine the glass bulb. If it is broken, pH readings will be incorrect.
If your readings are off by several pH values, the pH readings do not change when moved from one buffer solution to a buffer solution of different pH, the sensor was stored dry for an extended period of time, or the sensor’s response seems slow, the problem may be more serious. A method called “shocking” can be used to revive pH electrodes. To shock your pH Sensor, perform the following:
- Soak the pH Electrode for 4–8 hours in an HCl solution of 0.1 M–0.5 M.
- Rinse off the electrode and soak the tip in freshly prepared long term storage solution (recipe above) for 30–60 minutes.
- Rinse the electrode and test it with buffer solutions of known pH.
Occasionally, mold will grow in the pH 4 buffer/storage solution. Mold will not harm the electrode and can easily be removed using a mild detergent solution. Mold growth in the storage solution can be inhibited by adding a buffer preservative.
The stored pH calibration equation was determined in aqueous solutions. When determining the pH in organic media or non-aqueous solutions (less than 5% water), the conventional pH range of pH 0 to 14 is not valid because it is based on the dissociation behavior of water.
In applications involving non-aqueous solvents it is common to measure relative rather than absolute pH. Therefore, when doing a pH measurement in non-aqueous samples it is important to remember that the measurement will not give an absolute pH value. To measure quantitatively in non-aqueous solvents, prepare a calibration curve for the pH electrode with different samples that have a known composition corresponding to the conditions of the samples to be measured. This makes it possible to differentiate the different sample compositions during the measurement without having to quantify an absolute value during the measurement.
When testing in non-aqueous solutions, the electrode will lose its hydrated gel layer around the pH-sensitive membrane. To ensure that measurements can still be performed, take care to rehydrate the gel layer in an ion-rich aqueous solution between experiments.
For additional troubleshooting and FAQs, see www.vernier.com/til/3965
Repair Information
If you have followed the troubleshooting steps and are still having trouble with your Go Direct Glass-Body pH, contact Vernier Technical Support at support@vernier.com or call 888-837-6437. Support specialists will work with you to determine if the unit needs to be sent in for repair. At that time, a Return Merchandise Authorization (RMA) number will be issued and instructions will be communicated on how to return the unit for repair.
Accessories/Replacements
| Item | Order Code |
|---|---|
|
PH-SS |
|
|
PH-BUFCAP |
|
|
BTL |
|
|
GDX-GPH-BNC |
|
|
GDX-EA |
|
|
CB-USB-MICRO |
|
|
CB-USB-C-MICRO |
|
|
GDX-BAT-300 |
|
|
ETG |
Warranty
Warranty information for this product can be found on the Support tab at www.vernier.com/gdx-gph/#support
General warranty information can be found at www.vernier.com/warranty
Additionally, the warranty does not cover accidental breakage of the glass bulb of the pH electrode.
Disposal
When disposing of this electronic product, do not treat it as household waste. Its disposal is subject to regulations that vary by country and region. This item should be given to an applicable collection point for the recycling of electrical and electronic equipment. By ensuring that this product is disposed of correctly, you help prevent potential negative consequences on human health or on the environment. The recycling of materials will help to conserve natural resources. For more detailed information about recycling this product, contact your local city office or your disposal service.
Battery recycling information is available at www.call2recycle.org
Do not puncture or expose the battery to excessive heat or flame.
 The symbol, shown here, indicates that this product must not be disposed of in a standard waste container.
The symbol, shown here, indicates that this product must not be disposed of in a standard waste container.
Vernier Go Direct® Flat pH BNC Electrode
Order Code gdx-gph
 INSERT NEW BARCODE IN WORD
INSERT NEW BARCODE IN WORD
What's Included
- Go Direct Glass-Body pH
- Electrode Storage Bottle, containing pH 4/KCl solution
|
Bluetooth Connection |
USB Connection |
|
|
For full product documentation, see
www.vernier.com/manuals/gdx-gph
Note: Vernier products are for educational use only.
|
Federal Communication Commission Interference Statement
FCC Caution RF Exposure Warning IC Statement Industry Canada - Class B This digital apparatus does not exceed the Class B limits for radio noise emissions from digital apparatus as set out in the interference-causing equipment standard entitled “Digital Apparatus,” ICES-003 of Industry Canada. Operation is subject to the following two conditions: (1) this device may not cause interference, and (2) this device must accept any interference, including interference that may cause undesired operation of the device. To reduce potential radio interference to other users, the antenna type and its gain should be so chosen that the equivalent isotropically radiated power (e.i.r.p.) is not more than that permitted for successful communication. RF exposure warning: The equipment complies with RF exposure limits set forth for an uncontrolled environment. The antenna(s) used for this transmitter must not be co-located or operating in conjunction with any other antenna or transmitter.
Le présent appareil est conforme aux CNR d’Industrie Canada applicables aux appareils radio exempts de licence. L’exploitation est autorisée aux deux conditions suivantes : (1) l’appareil ne doit pas produire de brouillage, et (2) l’appareil doit accepter tout interférence radioélectrique, même si cela résulte à un brouillage susceptible d’en compromettre le fonctionnement. Avertissement d’exposition RF: L’équipement est conforme aux limites d’exposition aux RF établies pour un environnement non supervisé. L’antenne (s) utilisée pour ce transmetteur ne doit pas être jumelés ou fonctionner en conjonction avec toute autre antenne ou transmetteur.
Note: This product is a sensitive measurement device. For best results, use the cables that were provided. Keep the device away from electromagnetic noise sources, such as microwaves, monitors, electric motors, and appliances. |
Vernier Science Education
13979 S. W. Millikan Way • Beaverton, OR 97005-2886
Toll Free (888) 837-6437 • (503) 277-2299 • FAX (503) 277-2440
info@vernier.com • www.vernier.com
Rev. 2/15/2024
Go Direct, Vernier Graphical Analysis, LabQuest, and other marks shown are our trademarks or registered trademarks in the United States.
All other marks not owned by us that appear herein are the property of their respective owners, who may or may not be affiliated with, connected to, or sponsored by us.
The Bluetooth® word mark and logos are registered trademarks owned by the Bluetooth SIG, Inc. and any use of such marks by Vernier Science Education is under license. Other trademarks and trade names are those of their respective owners.


 Printed on recycled paper
Printed on recycled paper
Contact Support
Fill out our online support form or call us toll-free at 1-888-837-6437.