Experiments
Here are experiments our science specialists have selected to support the IB* topic.
Acid-Base Titration
Experiment #7 from Advanced Chemistry with Vernier
In this experiment, you will
- Accurately conduct acid-base titrations.
- Determine the equivalence point of a strong acid-strong base titration.
- Determine the equivalence point of a weak acid-strong base titration.
- Calculate the molar concentrations of two acid solutions.
Titration Curves of Strong and Weak Acids and Bases
Experiment #23 from Chemistry with Vernier
In this experiment, you will
- Observe differences in shapes of titration curves when various strengths of acids and bases are combined.
- Learn about the function and selection of appropriate acid-base indicators.
- Learn how to interpret the shape of a titration curve.
Acid-Base Titration
Experiment #24 from Chemistry with Vernier
In this experiment, you will
- Use a pH Sensor to monitor changes in pH as sodium hydroxide solution is added to a hydrochloric acid solution.
- Plot a graph of pH vs. volume of sodium hydroxide solution added.
- Use the graph to determine the equivalence point of the titration.
- Use the results to calculate the concentration of the hydrochloric acid solution.
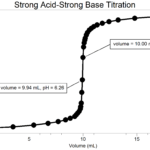
Acid-Base Titrations
Experiment #17 from Investigating Chemistry through Inquiry
In the Preliminary Activity, you will titrate a solution of the strong acid hydrochloric acid, HCl, with a solution of the strong base sodium hydroxide, NaOH. The concentration of the NaOH solution is given and you will determine the unknown concentration of the HCl.
After completing the Preliminary Activity, you will first use reference sources to find out more about acids, bases, and acid-base titrations before you choose and investigate a researchable question utilizing acid-base titrations.
- Educational Standard
- International Baccalaureate (IB)
- Subject
- Chemistry
- Section
- Core
- Topic
- 8. Acids and Bases
* The IB Diploma Program is an official program of the International Baccalaureate Organization (IBO) which authorizes schools to offer it. The material available here has been developed independently of the IBO and is not endorsed by it.
