Inspire Students to Make Meaning of Matter
Chemistry
Ready to get started with Go Direct?
Learn how to easily integrate Go Direct sensors into your classroom or lab.
Introductory Chemistry
Introduce your high school students to essential chemistry topics such as atomic structure, chemical bonding, stoichiometry, states of matter, chemical reactions, and fundamental principles of thermodynamics and kinetics using Vernier technology.
Forensics
Transform your classroom into a forensic science lab where students investigate fictitious crimes—and in the process, learn about physical and chemical properties, intermolecular forces, spectroscopy, and other key scientific concepts.
Food Chemistry
Students are more likely to authentically engage with science when they see chemistry concepts applied to the real world. Invite your students to examine the chemistry behind the snacks, drinks, and pantry items they consume on a regular basis through the lens of inquiry!

Introduce Your Students to Spectroscopy
Capable of connecting wirelessly or by USB, the Go Direct SpectroVis® Plus Spectrophotometer can easily collect a full wavelength spectrum (absorbance, percent transmittance, fluorescence, or emissions) in less than one second. Once the peak wavelength is determined, you can establish the concentration of a solution (Beer’s law) or monitor rates of reactions.
Software for Chemistry
Engage Students in Three-Dimensional, Hands-On Learning
Vernier Connections® powered by Penda deepens high school students’ understanding of scientific concepts and develops critical skills such as inquiry, problem solving, communication, and collaboration. Phenomenon-based, three-dimensional lessons combine the benefits of web‑based learning with hands-on investigation.

Collect, Graph, and Analyze Data in Real Time
Help students form critical connections between abstract scientific ideas and the real world. With the Vernier Graphical Analysis® app, students can visualize and interact with experiment data collected with nearly any Vernier sensor.
Capture, Analyze, and Share Spectrometer Data
Our free Vernier Spectral Analysis® app makes it easy to incorporate spectroscopy into your chemistry lab. Using the app, students can collect a full spectrum and explore topics such as Beer’s law, the rate of a chemical reaction, and equilibrium constants.
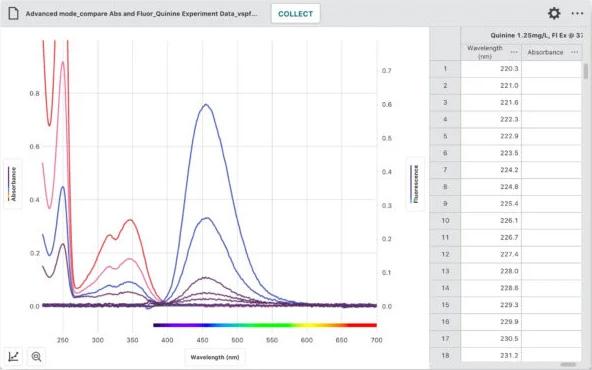

Top Three Topics That Students Miss in AP Chemistry Prep and How to Tackle Them
Certain topics, especially those in electrochemistry, thermodynamics, and kinetics, tend to get missed in AP Chemistry prep, either because they appear late in the curriculum, leaving little time for thorough coverage, or because their complexity can be daunting, leading some educators to skirt around them. We’re sharing some practical experiments from our lab books that can help you address these potential gaps.
Looking for more? Our Vernier Chemistry Investigations for Use with AP* Chemistry lab book offers 16 inquiry-based laboratory experiments aligned with the inquiry investigations published by the College Board.
Chemistry Packages
Ready to get started? Our ready-made packages feature top sensors tailored for different subjects and education levels. Need something specific? Our team is ready to help customize your order to meet your school’s exact needs.
See Our Data-Collection Technology in Action
Explore our expansive video library! Take a deeper dive into Vernier technology with our webinar recordings, get quick tips and practical demonstrations, and see how students can use sensors and software together.
*AP and Advanced Placement Program are registered trademarks of the College Entrance Examination Board, which was not involved in the production of and does not endorse this product.
We are scientists, educators, and your team.
As you implement data-collection technology into your teaching, we’re here to support you! Looking to learn more about our products or have questions about ordering? Reach out to our team at chemistry@vernier.com.
Are you a district or school administrator?
If you are considering an adoption or seeking training options, we are here to support you every step of the way. Contact our solutions team at k12outreach@vernier.com.