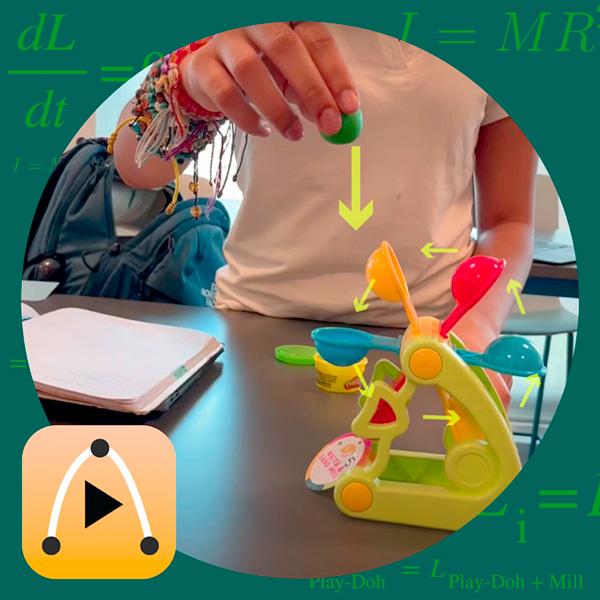
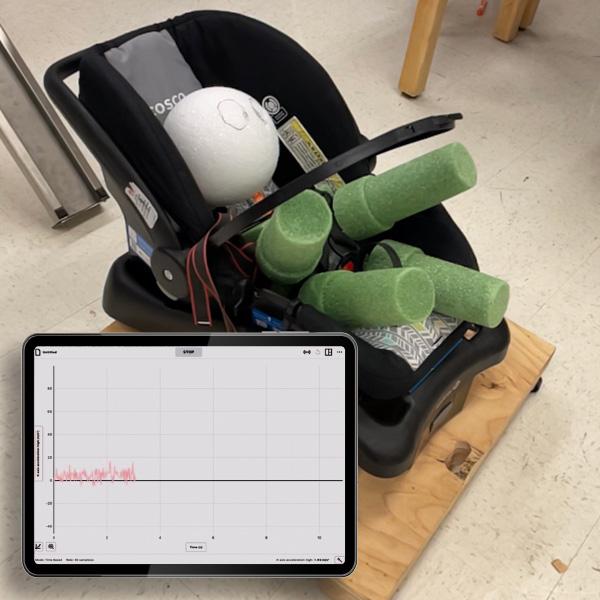
Get ready for National Chemistry Week with Vernier! This year, the American Chemical Society (ACS) is celebrating the chemistry behind photography and imaging. From traditional film cameras to cutting-edge medical imaging technologies, chemistry pla...